A Protein is Protecting You from Chronic Disease—And You’re Shutting It Off Eating THIS Way
Main Points
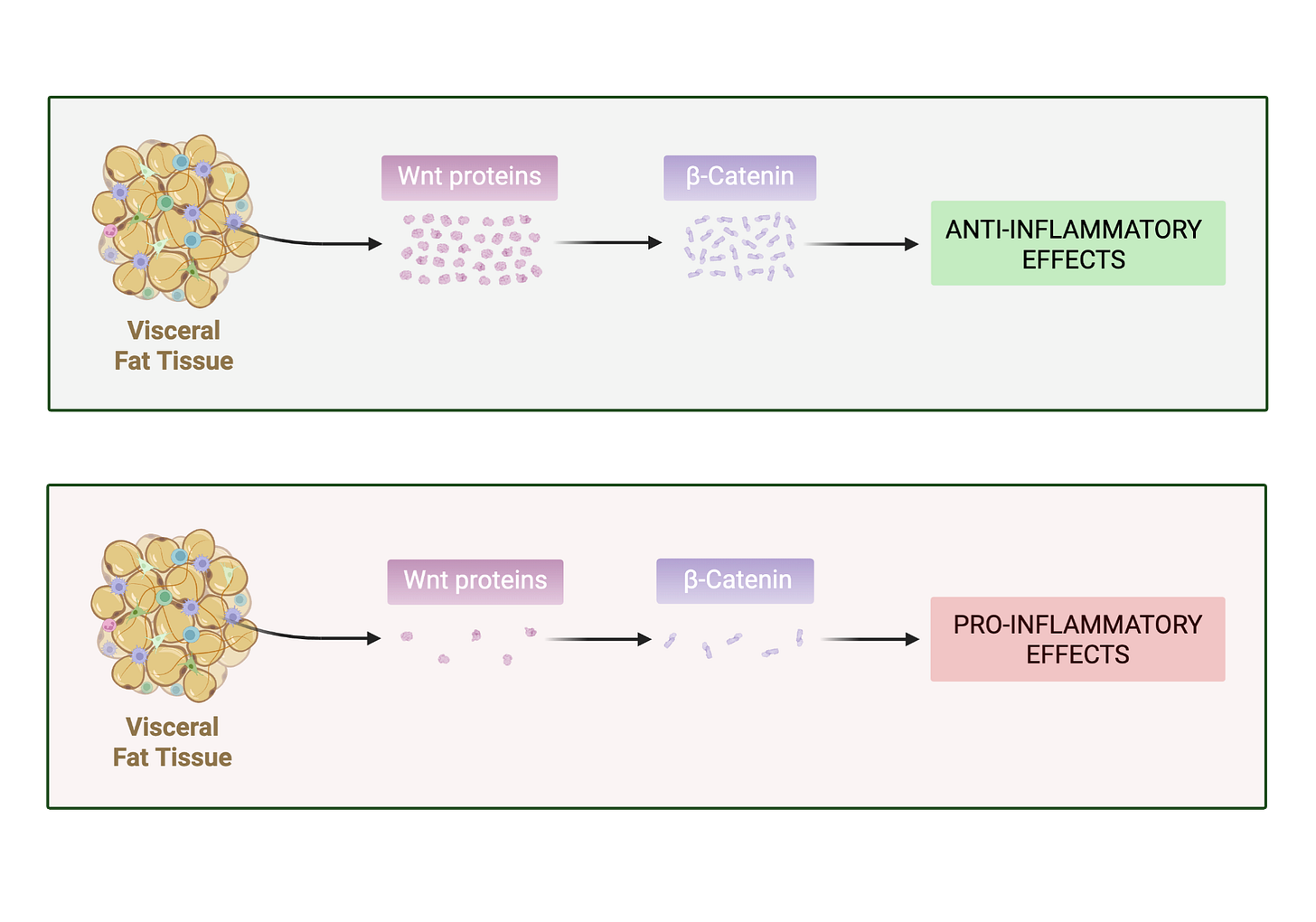
Visceral fat can influence immune cell inflammation by releasing Wnt proteins that activate beta-catenin, a key anti-inflammatory switch inside certain dendritic cells. A Western diet — high in sugar, saturated fats, and calories — reduces Wnt release and beta-catenin levels, pushing the immune system toward a more inflammatory state. Protecting beta-catenin means avoiding these dietary patterns and focusing on whole, nutrient-dense, less processed foods, minimizing added sugars, and reducing excess calorie intake.
● Another critical protein involved in health working through different mechanisms?
● Two supplements believed to improve dendritic cell function and reduce inflammation
● The loss of beta-catenin on insulin resistance and consequences
All of that is included in the complete analysis, along with access to a private podcast, live sessions with me, a library of articles and videos, and much more as a Physionic Insider :
Have you ever heard of a protein called beta-catenin?
Probably not — it’s a well-known player in cell biology and human physiology, but not something you often hear about in a clinical setting. Yet research shows it plays a powerful role in keeping inflammation in check — the very inflammation linked to diabetes, obesity, cardiovascular disease, cancer, and more. The surprising part? Your nutrition can severely reduce its levels, driving up inflammation and increasing disease risk.
Let’s break down what beta-catenin does, why it matters, and how you can keep it elevated in your body.
The Link Between Visceral Fat and Inflammation
This story starts in body fat — specifically, visceral fat. Unlike the fat just under your skin, visceral fat wraps around your organs and has been heavily linked to inflammation. Exactly how it drives inflammation has been the subject of intense research.
In this study, researchers examined how visceral fat influences immune cells. They used a technique called FACS (Fluorescence-Activated Cell Sorting) to separate out the different cells found inside visceral fat. Think of it as sorting people by facial features — but instead, they identified cells by molecular markers.
Among these cells were dendritic cells — a type of immune cell that acts as an “antigen-presenting cell.” Their job is to activate other immune cells and kick off inflammation by releasing signaling molecules called cytokines. While that’s good in the presence of infection, in the absence of a real threat it’s a recipe for unnecessary inflammation and potential tissue damage.
Beta-Catenin’s Anti-Inflammatory Role
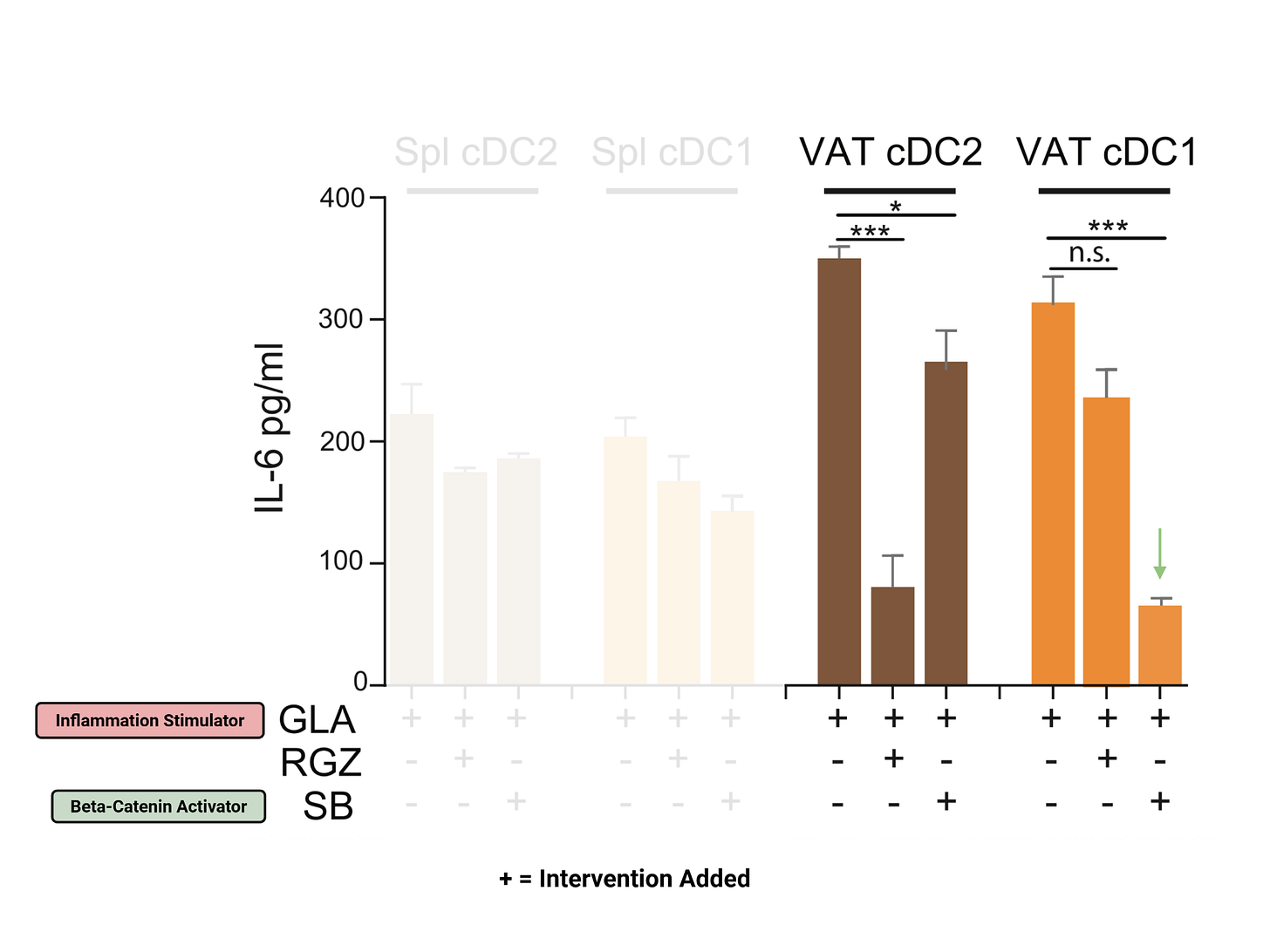
Within the dendritic cell population, there are subtypes — cDC1 and cDC2. The researchers found that cDC1 cells have much higher levels of beta-catenin than cDC2 cells.
Why is that important? Because when beta-catenin was activated, the production of interleukin-6 (IL-6) — a pro-inflammatory cytokine — dropped dramatically in cDC1 cells. This effect wasn’t nearly as strong in cDC2 cells, which makes sense given their lower beta-catenin levels. The findings were consistent across multiple measures, confirming beta-catenin’s inflammation-reducing role.
How Diet Can Switch Off Beta-Catenin
The researchers then looked at how beta-catenin is activated. One key activator is a signaling protein called Wnt10, which is released by visceral fat. When Wnt10 was applied to dendritic cells, IL-6 levels dropped just as they did with a direct beta-catenin activator, confirming that Wnt10 helps trigger beta-catenin’s anti-inflammatory action.
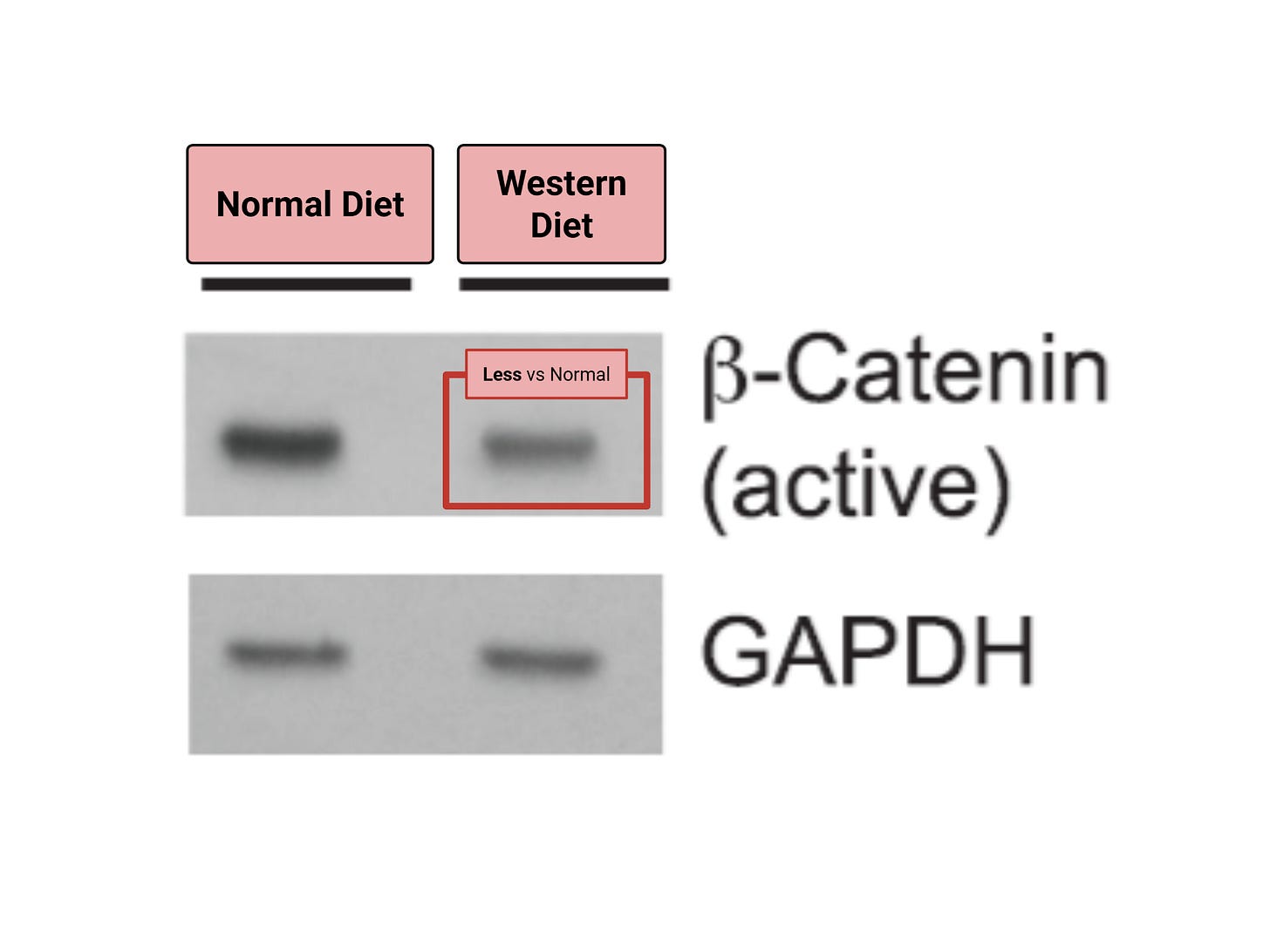
Here’s the catch: visceral fat doesn’t always release Wnt10. Diet plays a major role. In mice, a Western diet — high in sugar, saturated fats, and calories — significantly reduced Wnt10 release from visceral fat, and with it, beta-catenin levels. Lower beta-catenin meant higher inflammation potential.
What This Means for Health
While there’s limited direct human research linking beta-catenin, Wnt10, and inflammation from visceral fat, the overall picture aligns with extensive evidence that visceral fat promotes systemic inflammation. And inflammation, in turn, is strongly tied to chronic disease risk.
How to Protect Beta-Catenin Levels
The takeaway: avoid the dietary patterns that reduce beta-catenin and drive visceral fat–driven inflammation. That means steering clear of the typical Western diet and focusing on:
Eating more satiating, whole foods.
Cutting back on ultra-processed foods.
Reducing how often you eat out (most restaurant food is calorie-dense).
Limiting or eliminating sugar-sweetened drinks.
● Another critical protein involved in health working through different mechanisms?
● Two supplements believed to improve dendritic cell function and reduce inflammation
● The loss of beta-catenin on insulin resistance and consequences
All of that is included in the complete analysis, along with access to a private podcast, live sessions with me, a library of articles and videos, and much more as a Physionic Insider:
Main Points
Visceral fat can influence immune cell inflammation by releasing Wnt proteins that activate beta-catenin, a key anti-inflammatory switch inside certain dendritic cells. A Western diet — high in sugar, saturated fats, and calories — reduces Wnt release and beta-catenin levels, pushing the immune system toward a more inflammatory state. Protecting beta-catenin means avoiding these dietary patterns and focusing on whole, nutrient-dense, less processed foods, minimizing added sugars, and reducing excess calorie intake.
● Another critical protein involved in health working through different mechanisms?
● Two supplements believed to improve dendritic cell function and reduce inflammation
● The loss of beta-catenin on insulin resistance and consequences
All of that is included in the complete analysis, along with access to a private podcast, live sessions with me, a library of articles and videos, and much more as a Physionic Insider:
Dr. Nicolas Verhoeven, PhD / Physionic
References
[Study 506] Macdougall CE, Wood EG, Loschko J, et al. Visceral Adipose Tissue Immune Homeostasis Is Regulated by the Crosstalk between Adipocytes and Dendritic Cell Subsets. Cell Metabolism. 2018 Mar 6;27(3):588–601.e4. doi:10.1016/j.cmet.2018.02.007
Funding/Conflicts: Mixed Funding [Public: Public support came from the Medical Research Council (MRC) (MR/L002345/1) and the Medical College of Saint Bartholomew’s Hospital Trust, both of which are affiliated with publicly funded academic institutions. Non-profit funding included multiple grants from the British Heart Foundation (FS/13/49/30421, PG/16/79/32419, FS/14/66/31293), the Marie Curie / Cascade Fellowship (CF-2013-11-003-longhi); Non-Profit: Action Against Cancer and the Hilary Craft Foundation (PS8508). Additionally, one author was supported by a studentship from Bart’s and the London School of Medicine, a non-profit academic institution.] // No direct Conflicts of Interest
[A] Fontana L, Eagon JC, Trujillo ME, Scherer PE, Klein S. Visceral fat adipokine secretion is associated with systemic inflammation in obese humans. Diabetes. 2007;56(4):1010-1013. doi:10.2337/db06-1656
[B] Beasley LE, Koster A, Newman AB, et al. Inflammation and race and gender differences in computerized tomography-measured adipose depots. Obesity (Silver Spring). 2009;17(5):1062-1069. doi:10.1038/oby.2008.627